Ongoing, planned and completed studies
Over the past 20 years, pediatric investigational sites in our network have participated in actually 24 completed studies.
The indications are those commonly encountered in pediatric practices and outpatient clinics. Feasibility surveys were conducted in each case (black bars).
It was never possible to recruit all the children who were previously calculated during feasibility questionning to be possibly recruited. Nevertheless, 79% of the announced subjects could be included in vaccination studies. Only 1% oft he children dropped out In the case of the allergy and asthma studies, intensive screening was necessary in order to obtain the necessary number of subjects for the studies. Here in the middle 51% of announced subjects could be included in the studies, whereby a dropout of 21% was observed.
Scientific publications exist for many studies in which NETSTAP Centers participated. They are named below. Where the publications are missing, we mostly have official final reports by the sponsors.
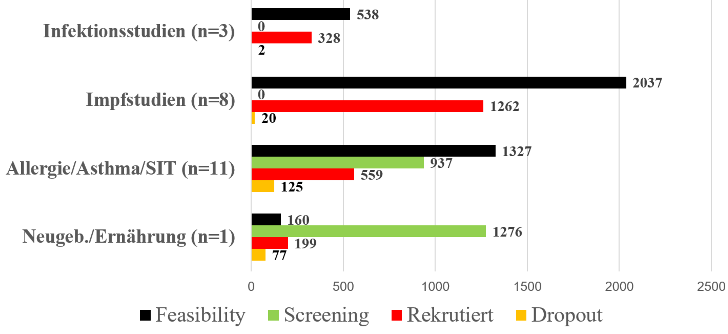
Here you will find an overview of our studies and their characteristics:
Ongoing studies
- Phase IIIb vacination study (2022-2027)
- Phase IIb vacination study (2022-2027)
- Phase III vacination study (2024-2026)
- SIT Studien im Rahmen der TAV (2024-2030)
- Phase II vacination study (2025-2026)
- Phase III vacination study (2025-2026)
Planned studies
-
Allergy prevention studies
-
further vaccine studies
-
spec. pediatric indication
Completed studies
-
- Nirsevimab for Prevention of Hospitalizations Due to RSV in Infants (2023)
-
Safety and Tolerability of V114 Pneumococcal Vaccine in Infants: A Phase 3 Study (2023)
- A Phase III, multicenter, randomized, double-blind, active comparator-controlled study to evaluate the safety, tolerability, and immunogenicity of V114 compared with PCV13 in healthy infants (PNEU-PED-EU-1) (2023)
- MenQuadfi_Pooled Naive toddlers_Poster_ECCMID2022_4April2022
- Side effects during subcutaneous immunotherapy (SCIT) in children with allergic diseases. (2017)
- 5-year SQ grass sublingual immunotherapy tablet asthma prevention (GAP) trail in children with grass pollen allergy (2017)
- Infant formula containing galacto-and fructo-oligosaccharides and Bifidobacterium breve M-16V supports adequate growth and tolerance in healthy infants in a randomised, controlled, double-blind, prospective, multicentre study (2016)
- Immunogenicity and tolerability of recombinant serogroup B meningococcal vaccine administered with or without routine infant vaccinations according to different immunization schedules: a randomized controlled trial (2012).
- High-dose sublingual immunotherapy with single-dose aqueous grass pollen extract in children is effective and safe: A double-blind, placebo-controlled study (2012)
- Safety an efficacy in adults and children of SQ-standardised mite allergen tablet for sublingual immunotherapy (2009)
- Safety of specific sublingual immunotherapy with SQ standardized grass allergen tablets in children (2007)
- Efficacy of sublingual swallow immunotherapy in children with severe grass pollen allergic symptoms (2004)